Chemical changes
Wednesday, December 21, 2011
Saturday, July 31, 2010
My Reflection
Reflection
When I saw the topic “chemical changes”, I thought it was a easy topic but after I went to research more, I realized it was hard and there are a lot of types of chemical changes, like electrolysis, decomposition, combination, cracking, photosynthesis, combustion and many many more. Luckily my mom was there to help me she took out five books about chemistry and told me that they would help me with my project, I started by finding out the types of chemical changes and then grouped them. Then using the textbooks my mom gave me, I did more background research, but most were hard and I had to rely on my mom to explain it to me. In the end, I finished the entire thing and now doing the report.
One question that occurred to me is “what is the difference between combustion and oxidation?” In my research, in oxidation, magnesium burns in air and the magnesium will be oxidised by the oxygen, but yet it also combustion describes the chemical reaction where the magnesium reacts with oxygen in an exothermic reaction. So it makes me wonder what is the difference between these two chemical changes, combustion or oxidation? Another question is photosynthesis is endothermic which means it takes in heat energy for photosynthesis to occur, but photosynthesis takes place when the chloroplast takes in sunlight. So why is photosynthesis an endothermic reaction when it takes in sunlight??Another question is “Why is respiration considered a chemical reaction that involves heat?”, respiration is only the exchange of gases in the lungs. Another question is will a strong acid be neutralized by a weak acid ? and vice versa.
I encountered a few difficulties in the project and that is understanding everything in the book or in the web. Most of the information were very hard to understand because those information must be for more advanced students. Other difficulties like the information from different websites were different therefore making me confused. I solved it by checking different websites to make sure my information was correct. Another difficulty is time and because the examination was a week away, we had to both study and do this project( this project is twenty percent of the mastery review), so we had to plan our time very well or else the project and the studying would not be completed. Another difficulty is that there are so many types of chemical change that it is so difficult to put everything into the glog, so I just took out a few main ones and just researched on them
From this I learnt how to plan my time well, paraphrase paragraphs that were too long or to prevent plagiarism .I also feel that this project is very good because a lot of us will learn new things, it also increase our information on our topic so that we are more prepared on that certain topic during the exam. This project also is good because we have to do our own reflection as reflection is important or you might as well copy paste and not learn anything.
Friday, July 30, 2010
References
Glossary......
CHEMICAL CHANGES INVOLVING HEAT

Respiration
CHEMICAL CHANGES INVOLVING LIGHT

Photosynthesis
The reaction of photosynthesis utilizes six carbon dioxide molecules and six water molecules to produce one sugar molecule (glucose) and six oxygen molecules.
Photosynthesis is the process by which plants and some single-celled organisms produce sugars from sunlight and carbon dioxide. These sugars then become the food used in cellular respiration. In plants, photosynthesis occurs in specialized structures known as chloroplasts.
CHEMICAL CHANGES INVOLVING HEAT
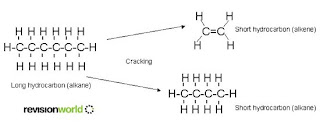
Cracking